G.Patton
Expert
- Joined
- Jul 5, 2021
- Messages
- 2,412
- Solutions
- 3
- Reaction score
- 2,381
- Points
- 113
- Deals
- 1
Crack is the free base of cocaine (not bonded with acid). It gives 4 major by-products under heating, whose structures are presented in picture below.
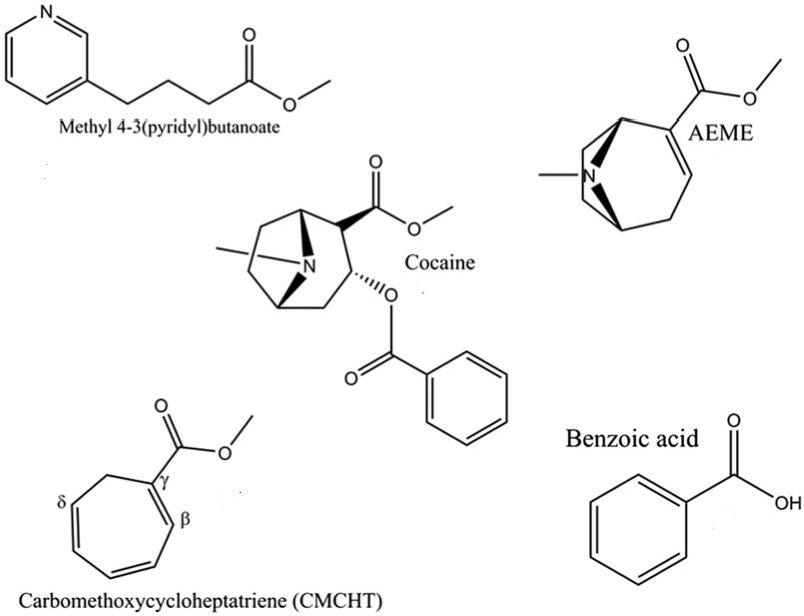
These four major by-products are hydroxyecgonine methyl ester (AEME), benzoic acid, carbomethoxycycloheptatriene and methyl 4-(3-pyridinyl) butanoate.
AEME.
It causes toxic encephalopathy, the most toxic by-product.
Carbomethoxycycloheptatriene.
Its structure is similar to 1-methoxy-1,3,5-cycloheptatriene’s one. It is known to be a painful chemical warfare agent. And, although it does not have the same toxicity, it causes extremely unpleasant sensations in places of contact with the skin and mucous membranes, forms long-lasting ulcers on the lips, tongue and fingers.
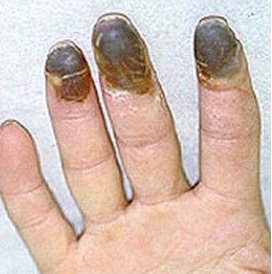
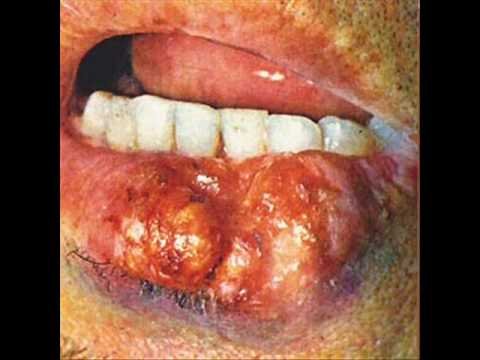
Benzoic acid.
The most innocuous side product among substances mentioned above, but also is useless.
Methyl 4-(3-pyridinyl)butanoate.
The metabolic pathway is not investigated completely. This substance is known to persist in organism for up to 7 days and serves as a marker of the crack use by smoking.
Literature:
These four major by-products are hydroxyecgonine methyl ester (AEME), benzoic acid, carbomethoxycycloheptatriene and methyl 4-(3-pyridinyl) butanoate.
AEME.
It causes toxic encephalopathy, the most toxic by-product.
Carbomethoxycycloheptatriene.
Its structure is similar to 1-methoxy-1,3,5-cycloheptatriene’s one. It is known to be a painful chemical warfare agent. And, although it does not have the same toxicity, it causes extremely unpleasant sensations in places of contact with the skin and mucous membranes, forms long-lasting ulcers on the lips, tongue and fingers.
Benzoic acid.
The most innocuous side product among substances mentioned above, but also is useless.
Methyl 4-(3-pyridinyl)butanoate.
The metabolic pathway is not investigated completely. This substance is known to persist in organism for up to 7 days and serves as a marker of the crack use by smoking.
Studies have been showed the direct dependence of the amount of the side products on temperature: the higher temperature, the more side toxic products.
For example, at the temperature of 120 C, only 7% of the crack decomposes into side products. At temperature of 170 C, 220 C and 270 C, 27%, 38% and 63% of crack decomposes respectively. At the temperature above 300 C, almost all cracks will be decomposed by side products. AEME does not form at the temperature below 220 C.
Free base of cocaine sublimes at the temperature of 90-98 C. Therefore, heat the crack solely in a water bath (until the water boils away, the temperature does not rise above 100 C).
For example, at the temperature of 120 C, only 7% of the crack decomposes into side products. At temperature of 170 C, 220 C and 270 C, 27%, 38% and 63% of crack decomposes respectively. At the temperature above 300 C, almost all cracks will be decomposed by side products. AEME does not form at the temperature below 220 C.
Free base of cocaine sublimes at the temperature of 90-98 C. Therefore, heat the crack solely in a water bath (until the water boils away, the temperature does not rise above 100 C).
Literature:
Y. Nakahara, A. Ishigami. Inhalation efficiency of free-base cocaine by pyrolysis of crack and cocaine hydrochloride. J. Anal. Toxicol. 1991, 15, 105. [16] J.A. Fournier, J.B. Paine, J.I. Seeman, D.W. Armstrong, X.H. Chen. Thermal pathways for the transfer of amines, including nicotine, to the gas phase and aerosols. Heterocycles. 2001, 55, 59.
S.W. Toennes, A.S. Fandino, G. Kauert. Gas chromatographic-mass spectrometric detection of anhydroecgonine methyl ester (Methylecgonidine) in human serum as evidence of recent smoking of crack. J. Chrom. B. 1999, 735, 127.
K.J. Riley, N.T. Lu, J.E. Meeker, P. Lo, N. Fortner, B.G. Taylor. Monitoring the crack epidemic through urine testing: Establishment of routine detection methods. Addict. Biol. 2001, 6, 83.
H.J. Liberty, B.D. Johnson, N. Fortner, D. Randolph. Detecting crack and other cocaine use with fast patches. Addict. Biol. 2003, 8, 191.
R.J. Lewis, R.D. Johnson, M.K. Angier, R.M. Ritter. Determination of cocaine, its metabolites, pyrolysis products, and ethanol adducts in postmortem fluids and tissues using Zymark® automated solidphase extraction and gas chromatography–mass spectrometry. J. Chrom. B. 2004, 806, 141.
A.L. Myers, H.E. Williams, J.C. Kraner, P.S. Callery. Identification of anhydroecgonine ethyl ester in the urine of a drug overdose victim. J. Forensic Sci. 2005, 50, 1481.
P.S. Cardona, A.K. Chaturvedi, J.W. Soper, D.V. Canfield. Simultaneous analyses of cocaine, cocaethylene, and their possible metabolic and pyrolytic products. Forensic Sci. Int. 2006, 157, 46.
T. Kraemer, L.D. Paul. Bioanalytical procedures for determination of drugs of abuse in blood. Anal. Bioanal. Chem. 2007, 388, 1415.
S.W. Toennes, A.S. Fandino, G. Kauert. Gas chromatographic-mass spectrometric detection of anhydroecgonine methyl ester (Methylecgonidine) in human serum as evidence of recent smoking of crack. J. Chrom. B. 1999, 735, 127.
K.J. Riley, N.T. Lu, J.E. Meeker, P. Lo, N. Fortner, B.G. Taylor. Monitoring the crack epidemic through urine testing: Establishment of routine detection methods. Addict. Biol. 2001, 6, 83.
H.J. Liberty, B.D. Johnson, N. Fortner, D. Randolph. Detecting crack and other cocaine use with fast patches. Addict. Biol. 2003, 8, 191.
R.J. Lewis, R.D. Johnson, M.K. Angier, R.M. Ritter. Determination of cocaine, its metabolites, pyrolysis products, and ethanol adducts in postmortem fluids and tissues using Zymark® automated solidphase extraction and gas chromatography–mass spectrometry. J. Chrom. B. 2004, 806, 141.
A.L. Myers, H.E. Williams, J.C. Kraner, P.S. Callery. Identification of anhydroecgonine ethyl ester in the urine of a drug overdose victim. J. Forensic Sci. 2005, 50, 1481.
P.S. Cardona, A.K. Chaturvedi, J.W. Soper, D.V. Canfield. Simultaneous analyses of cocaine, cocaethylene, and their possible metabolic and pyrolytic products. Forensic Sci. Int. 2006, 157, 46.
T. Kraemer, L.D. Paul. Bioanalytical procedures for determination of drugs of abuse in blood. Anal. Bioanal. Chem. 2007, 388, 1415.
Last edited:
