G.Patton
Expert
- Joined
- Jul 5, 2021
- Messages
- 2,596
- Solutions
- 3
- Reaction score
- 2,623
- Points
- 113
- Deals
- 1
Introduction
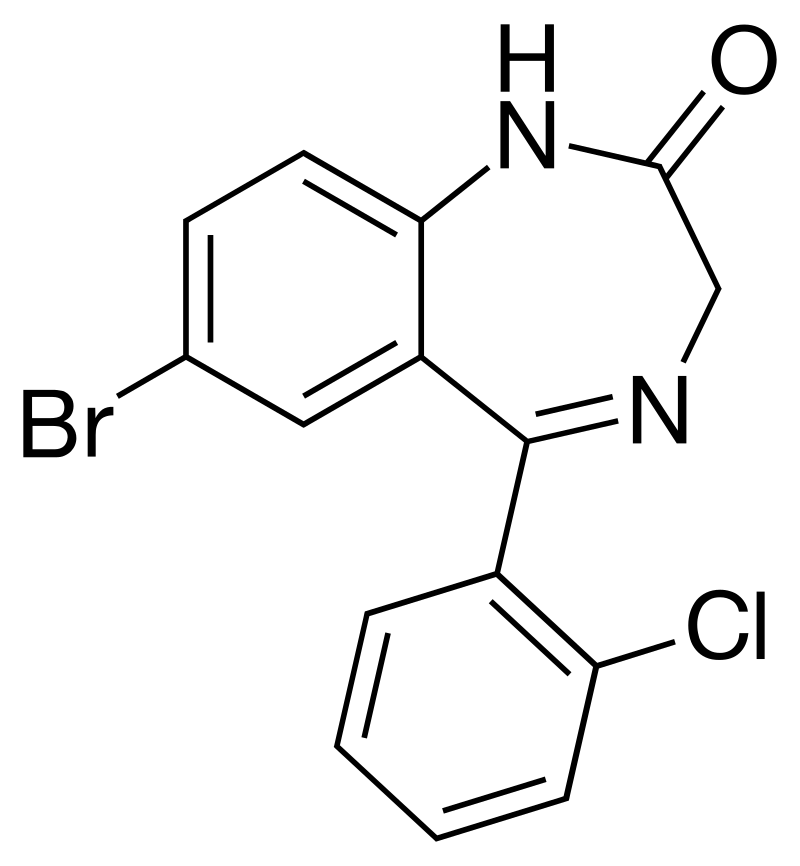
I would love to represent to BB public classical Phenazepam (Bromdihydrochlorphenylbenzodiazepine; 7-Bromo-5-(2-chlorophenyl)-1,3-dihydro-2H-1,4-benzodiazepin-2-one; Fenazepam; BD 98) synthesis from p-bromoaniline and o-chlorobenzoic acid. This synthesis route assumes the laboratory skills and experience of the chemist in a variety of laboratory techniques. At the same time, this method does not require the use of exotic reagents or harsh methods of processing substances.
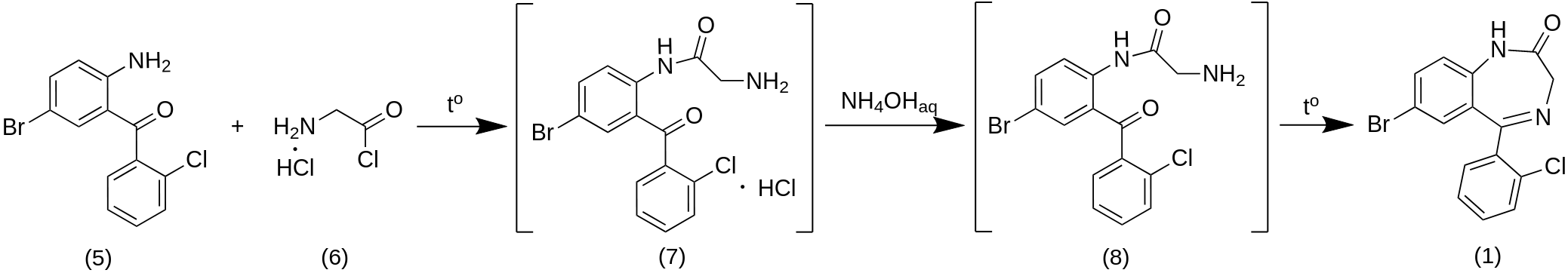
Acylation of p-bromoaniline (2) with o-chlorobenzoic acid chloride (3) in the presence of a catalyst - zinc chloride (ZnCl2) -gives 2-(o-chlorobenzoylamino)-5-bromo-2-chlorobenzophenone (4), which is hydrolyzed without isolation with an aqueous solution of H2SO4 to 2-amino-5-bromo-2-chlorobenzophenone (5). 2-Amino-5-bromo-2-chlorobenzophenone (5) is converted into Phenazepam without isolation of intermediate substances as follows: 5 is acylated with aminoacetic acid chloride hydrochloride (6) in chloroform, the resulting 2-(aminomethylcarbonylamino)-5-bromo-2-chlorobenzophenone hydrochloride (7) is converted by the action of aqueous NH3 to base (8), which is thermally cyclized to Phenazepam. 6 is prepared by treating PCl5 glycyrne in chloroform.
White or white crystalline powder with a cream shade, practically insoluble in water, hardly soluble in ether, alcohol, chloroform, soluble in benzene, toluene, dioxane, dimethylformamide;
Boiling Point: 493.0±45.0 °C/760 mm HgAcylation of p-bromoaniline (2) with o-chlorobenzoic acid chloride (3) in the presence of a catalyst - zinc chloride (ZnCl2) -gives 2-(o-chlorobenzoylamino)-5-bromo-2-chlorobenzophenone (4), which is hydrolyzed without isolation with an aqueous solution of H2SO4 to 2-amino-5-bromo-2-chlorobenzophenone (5). 2-Amino-5-bromo-2-chlorobenzophenone (5) is converted into Phenazepam without isolation of intermediate substances as follows: 5 is acylated with aminoacetic acid chloride hydrochloride (6) in chloroform, the resulting 2-(aminomethylcarbonylamino)-5-bromo-2-chlorobenzophenone hydrochloride (7) is converted by the action of aqueous NH3 to base (8), which is thermally cyclized to Phenazepam. 6 is prepared by treating PCl5 glycyrne in chloroform.
White or white crystalline powder with a cream shade, practically insoluble in water, hardly soluble in ether, alcohol, chloroform, soluble in benzene, toluene, dioxane, dimethylformamide;
Melting Point: 220-222 °C
Molecular Weight: 349.61 g/mol
Density: 1.6±0.1 g/cm3 (20 °C)
Index of Refraction: 1.692
CAS Number: 51753-57-2
Equipment and glassware:
5 L three necked round bottom flasks;5 L round bottom flask;
Laboratory grade thermometer (0 °C to 200 °C) with flask adapter;
Water bath and Ice;
Heater;
Reflux condenser;
Magnetic stirrer;
Drip funnel 1 L;
5 L x2; 1 L x2; 500 ml x2; 100 ml x2 Beakers;
Conventional large funnels x2 (~20-25 cm);
Buchner flask 5 L and funnel (large);
Water-jet aspirator;
Vacuum desiccator;
Measuring cylinders 100 and 1000 ml;
Glass rod;
Laboratory scale (1 - 1000 g is suitable);
Distillation apparatus with Vigreux column;
Retort stand and clamp for securing apparatus;
Receiver flask (2-3 L);
Filter paper;
Separatory funnel 5 L.
Reagents:
276 g p-Bromoaniline (2);672 g o-Chlorobenzoic acid (3);
264 g Zinc chloride (ZnCl2);
0.6 L of 14% aq. Hydrochloric acid (HCl) ;
3 L Distilled water;
0.6 L 72% aq. Sulfuric acid (H2SO4);
500 ml 20% aq. Sodium hydroxide (NaOH);
2 L Chloroform;
~3 l Toluene;
496 g Hydrochloride of aminoacetic acid chloride (6);
~500 ml Ammonia solution NH4OH aq.;
1 L Ethanol (96-98%) (EtOH).
2-amino-5-bromo-2-chlorobenzophenone (5)
A mixture of 276 g of p-bromoaniline (2) and 672 g of o-chlorobenzoic acid (3) in 5 L three necked round bottom flasks with thermometer are heated for 1 hour at 100-110 °C with a reflux condenser, the temperature is raised to 150-160 °C, 264 g of ZnCl2 are added, heated for 2 hours at 190-198 °C and cool. 0.6 L of 14% hydrochloric acid (HCl) is added to the mass via drip funnel, heated to 100 °C, the aqueous layer is separated, the organic layer is washed by boiling with 0.3 l of distilled water. The washing procedure is repeated three times. After washing, 0.6 L of 72% sulfuric acid (H2SO4) is added to the residue, heated for 2 hours at 160 °C and poured into a 5 L baker with a mixture of ice and water. The precipitate is filtered in a large Buchner flask and funnel, washed with distilled water, stirred for 1 hour with a 20% aqueous solution of NaOH, filtered, washed in a separatory funnel with water until pH 7 and dried on air. The yield of technical grade 2-amino-5-bromo-2-chlorobenzophenone (5) 228 g with a basic substance content of 70%.
7-Bromo-5-(2-chlorophenyl)-1,3-dihydro-2H-1,4-benzodiazepin-2-one (Fenzepam) (1)
A solution of 850 g of 2-amino-5-bromo-2-chlorobenzophenone (5) in 2 L of chloroform is dried in 5 L round bottom flask by azeotropic distillation of chloroform and water mixture, 496 g of hydrochloride of aminoacetic acid chloride (6) is added, boiled for 3 hours with a reflux condenser and cooled to room temperature. To the mixture is slowly poured a dilute aqueous ammonia solution to reach pH 8, the organic layer is washed with water in separatory funnel and evaporated to dryness in rotavap apparatus. To the residue is poured 2 L of toluene, heated to boiling, the azeotropic mixture of toluene and water is distilled off, cooled and filtered in a Buchner flask. Technical grade Phenazepam is crystallized (1:60) from toluene in 5 L baker, filtered, the precipitate is washed with toluene, alcohol (EtOH) and dried at 100-110 °C. Yield 250 g (26%).
Last edited:
