William D.
Expert
- Joined
- Jul 19, 2021
- Messages
- 1,055
- Reaction score
- 1,324
- Points
- 113
3-[2-(Dimethylamino)-2-oxoacetyl]-1H-indol-4-yl Acetate.
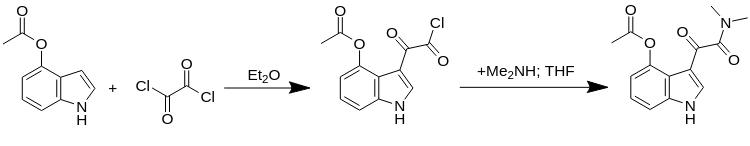 1. Into a round-bottomed flask was equipped with an overhead stirrer, temperature controller, and dropping funnel was added 1H-indol-4-yl acetate (50.1 g, 285 mmol, 1 equiv) and anhyd Et2O (700 ml).
1. Into a round-bottomed flask was equipped with an overhead stirrer, temperature controller, and dropping funnel was added 1H-indol-4-yl acetate (50.1 g, 285 mmol, 1 equiv) and anhyd Et2O (700 ml).
2. The suspension was stirred for 10 min and then cooled to 0 *C in an ice-water bath for 30 min.
3. The dropping funnel was charged with a solution of oxalyl chloride (37.1 ml, 428 mmol,1.5 equiv) in Et2O (60 ml).
4. The oxalyl chloride solution was added dropwise at a rate sufficient to keep the temperature at or below 5 *C, to minimize formation of dimer and other possible by-product.
5. As the addition progressed, a yellow slurry was formed and when the addition was completed, the mixture was stirred for 4 h.
6. After this time, heptane (400 ml) was added and the mixture stirred for 30 min at 0 *C.
7. The yellow solid obtained was quickly filtered and rinsed successively with heptane (2x300 ml), which was quickly dissolved in THF (500 ml) and cooled to 0 *C.
8. A 2.0 M solution of dimethylamine in THF (175 ml) was added dropwise at a rate sufficient to maintain temperature below 5 *C in order to minimize side reactions.
9. After the addition was complete, pyridine (46 ml) in of THF (100 ml) was added dropwise and the mixture was stirred well for 60 min.
10. Heptane (600 ml) was added, and the flask contents were suction filtered via a Büchner funnel.
11. The filtered residue was transferred into a roundbottomed flask and deionized H2O (1000 ml) was added, stirred for 30 min, and filtered via Büchner funnel.
12. The off-white solid was triturated sequentially for 40 min in EtOAc (600 ml) and heptane (400 ml).
13. The slurry was filtered via Büchner funnel and the solid was dried in an oven at 40 *C overnight to afford 6 as a light-yellow solid; yield: 66.1 g (81%).
3-[2-(Dimethylamino)ethyl]-1H-indol-4-ol (Psilocin).
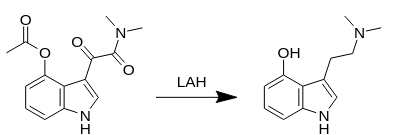 Procedure A:
Procedure A:
1. . Into a round-bottomed flask was equipped with an overhead stirrer, temperature controller, and a dropping funnel was added 3-[2-(dimethylamino)-2-oxoacetyl]-1H-indol-4-yl acetate (31.5 g, 115 mmol) and 2-CH3-THF (1000 ml).
2. The flask was immersed in an ice-bath at 0 *C and a solution of 2.3 M LiAlH4 in 2-CH3-THF (140 ml, 322 mmol) was added through the dropping funnel.
3. The dropping funnel was rinsed with additional 2-CH3-THF (20 ml). The LiAlH4 solution was added dropwise at a rate to maintain a temperature below 20 *C.
4. After the addition, the ice-water bath was removed and the mixture stirred for 30 min.
5. The light-yellow solution was heated to reflux (80 *C) with a heating mantle and became ivory-colored after 3 h.
6. Accumulation of yellow solids was observed on the sides of the round-bottomed flask.
7. The heating mantle was removed, and the flask was allowed to cool to 50 *C.
8. The flask was again chilled to 20 *C.
9. The reaction was quenched by sequential addition of 3 drops of aq. 1 M NaOH and 3 drops of deionized H2O.
10. The mixture was diluted with THF (500 ml) and stirred for 20 min.
11. The mixture was filtered via Büchner funnel and the filtrate was kept under N 2.
12. The filter cake was quickly reslurried with 200 ml of [10% solution of (7% ammonia in MeOH) in CH2Cl2] and THF (500 ml).
13. The filtrates were then combined and concentrated to give a green solid.
14. The solid was triturated with 1:1 EtOAc/heptane (50 ml), then filtered via Büchner funnel.
15. The dark green solid was dried in an oven at 40 *C overnight to provide dry psilocin as a dark green solid; yield: 20.7 g (91%); mp 167–169 *C.
Procedure B:
1. The reduction step was carried out using essentially the same protocol described in Procedure A with 3-[2-(Dimethylamino)-2-oxoacetyl]-1H-indol-4-yl Acetate (40.21 g, 135.2 mmol) and 2.3 M LiAlH4 in 2-CH3-THF (188.1 ml, 432.5 mmol).
2. The reaction was quenched by dropwise addition of THF/H2O (27:100, 50 ml) at a rate that kept the temperature below 30 *C.
3. Anhyd Na2SO4 (100 g) was added, followed by silica gel (50 g) and DCM (400 ml).
4. The mixture was stirred for 10 min and was filtered via Büchner funnel.
5. The filter cake was washed with DCM/CH3OH mixture (9:1, 1500 ml).
6. The filtrates were then combined and concentrated to give a light green solid.
7. The solid was triturated with 1:1 EtOAc/heptane (50 ml), then filtered via Büchner funnel.
8. The off-white solid was dried in an oven at 40 *C overnight to provide dry psilocin as an off-white solid; yield: 21.6 g (77%).
2. The suspension was stirred for 10 min and then cooled to 0 *C in an ice-water bath for 30 min.
3. The dropping funnel was charged with a solution of oxalyl chloride (37.1 ml, 428 mmol,1.5 equiv) in Et2O (60 ml).
4. The oxalyl chloride solution was added dropwise at a rate sufficient to keep the temperature at or below 5 *C, to minimize formation of dimer and other possible by-product.
5. As the addition progressed, a yellow slurry was formed and when the addition was completed, the mixture was stirred for 4 h.
6. After this time, heptane (400 ml) was added and the mixture stirred for 30 min at 0 *C.
7. The yellow solid obtained was quickly filtered and rinsed successively with heptane (2x300 ml), which was quickly dissolved in THF (500 ml) and cooled to 0 *C.
8. A 2.0 M solution of dimethylamine in THF (175 ml) was added dropwise at a rate sufficient to maintain temperature below 5 *C in order to minimize side reactions.
9. After the addition was complete, pyridine (46 ml) in of THF (100 ml) was added dropwise and the mixture was stirred well for 60 min.
10. Heptane (600 ml) was added, and the flask contents were suction filtered via a Büchner funnel.
11. The filtered residue was transferred into a roundbottomed flask and deionized H2O (1000 ml) was added, stirred for 30 min, and filtered via Büchner funnel.
12. The off-white solid was triturated sequentially for 40 min in EtOAc (600 ml) and heptane (400 ml).
13. The slurry was filtered via Büchner funnel and the solid was dried in an oven at 40 *C overnight to afford 6 as a light-yellow solid; yield: 66.1 g (81%).
3-[2-(Dimethylamino)ethyl]-1H-indol-4-ol (Psilocin).
1. . Into a round-bottomed flask was equipped with an overhead stirrer, temperature controller, and a dropping funnel was added 3-[2-(dimethylamino)-2-oxoacetyl]-1H-indol-4-yl acetate (31.5 g, 115 mmol) and 2-CH3-THF (1000 ml).
2. The flask was immersed in an ice-bath at 0 *C and a solution of 2.3 M LiAlH4 in 2-CH3-THF (140 ml, 322 mmol) was added through the dropping funnel.
3. The dropping funnel was rinsed with additional 2-CH3-THF (20 ml). The LiAlH4 solution was added dropwise at a rate to maintain a temperature below 20 *C.
4. After the addition, the ice-water bath was removed and the mixture stirred for 30 min.
5. The light-yellow solution was heated to reflux (80 *C) with a heating mantle and became ivory-colored after 3 h.
6. Accumulation of yellow solids was observed on the sides of the round-bottomed flask.
7. The heating mantle was removed, and the flask was allowed to cool to 50 *C.
8. The flask was again chilled to 20 *C.
9. The reaction was quenched by sequential addition of 3 drops of aq. 1 M NaOH and 3 drops of deionized H2O.
10. The mixture was diluted with THF (500 ml) and stirred for 20 min.
11. The mixture was filtered via Büchner funnel and the filtrate was kept under N 2.
12. The filter cake was quickly reslurried with 200 ml of [10% solution of (7% ammonia in MeOH) in CH2Cl2] and THF (500 ml).
13. The filtrates were then combined and concentrated to give a green solid.
14. The solid was triturated with 1:1 EtOAc/heptane (50 ml), then filtered via Büchner funnel.
15. The dark green solid was dried in an oven at 40 *C overnight to provide dry psilocin as a dark green solid; yield: 20.7 g (91%); mp 167–169 *C.
Procedure B:
1. The reduction step was carried out using essentially the same protocol described in Procedure A with 3-[2-(Dimethylamino)-2-oxoacetyl]-1H-indol-4-yl Acetate (40.21 g, 135.2 mmol) and 2.3 M LiAlH4 in 2-CH3-THF (188.1 ml, 432.5 mmol).
2. The reaction was quenched by dropwise addition of THF/H2O (27:100, 50 ml) at a rate that kept the temperature below 30 *C.
3. Anhyd Na2SO4 (100 g) was added, followed by silica gel (50 g) and DCM (400 ml).
4. The mixture was stirred for 10 min and was filtered via Büchner funnel.
5. The filter cake was washed with DCM/CH3OH mixture (9:1, 1500 ml).
6. The filtrates were then combined and concentrated to give a light green solid.
7. The solid was triturated with 1:1 EtOAc/heptane (50 ml), then filtered via Büchner funnel.
8. The off-white solid was dried in an oven at 40 *C overnight to provide dry psilocin as an off-white solid; yield: 21.6 g (77%).
Last edited by a moderator:
