- Language
- 🇬🇧
- Joined
- Jul 6, 2021
- Messages
- 328
- Reaction score
- 343
- Points
- 63
Dextroamphetamine (D-AMP) belongs to a class of central nervous system stimulants. The food and Drug Administration of the USA classifies it as a schedule II substance with a high potential for abuse. Medications with immediate and delayed release of amphetamine are approved by FDA for treatment of ADHD and narcolepsy both in adults and children. Dextroamphetamine is different from its reflection – levoamphetamine – because it has a more pronounced stimulation of the nervous system due to dopamine, and less - due to norepinephrine.
Synthesis of Dextroamphetamine from P2P
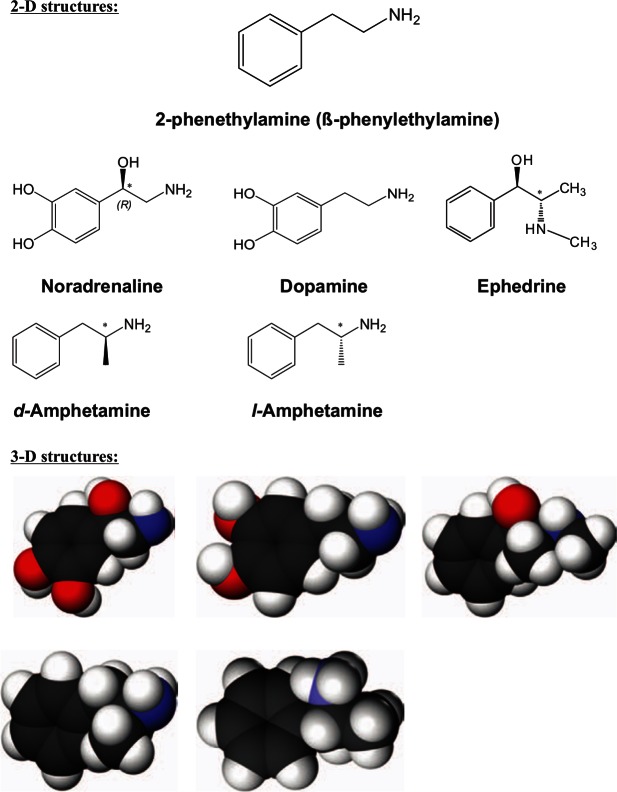
Physico-chemical properties and forms of the substance.
Fundamentally, dextroamphetamine, as well as amphetamine racemate, has the appearance of a colorless volatile oily liquid with a characteristic "fishy" smell and is well-soluble in organic solvents. Its boiling point is 200-203 °C. The enantiomer can have a powdery appearance with the formation of small "stones" of white or light yellow/cream color, with a weak smell, weakly hygroscopic, partially soluble in water when it is in the form of a tablet. It is most often found in the form of sulfate, hydrochloride, sucrate and very rarely in the form of phosphate. As a rule, dextroamphetamine is part of various medications such as «Aderral» and «Dexedrine», where it is found in the form of sucrate and sulfate in concentrations of 25% of the total volume. It also is part of «Vyvanse», «Elvanse», «Tyvense», where it is in the proactive state of dimesylate lysdexamphetamine; fermentation with l-lysine, metabolizes it into the active form of d-amphetamine; «Dexedrine». Other trade names of drugs include: Dexedrine sulphate, Afatin, d-Amfetasul, Domafate Obesedrin, Dexten, Maxiton, Sympamin, Simpamina-D, Albemap, Dadex, Ardex, Dexalone, Amsustain, Betafedrina, d-Betaphedrine, Diocurb, Dextrostat.
Pharmacokinetics.
When administered, amphetamine is metabolized by hydroxylation, where the main cytochrome is CYP2D6, beta-hydroxylation (DBH) and oxidative deamination (FMO3); there are other enzymes that are less involved in metabolism of amphetamine, such as butyrate-CoA ligase and glycine-N-acyltransferase. Bioavailability (with unchanged gastrointestinal pH) reaches 70-75% for racemate and up to 80-83% for dextroamphetamine. The binding with plasma proteins reaches 15-30% according to various sources in healthy people, the distribution volume is moderate, the substance actively passes through the blood-brain barrier with a variable coefficient of 17p. T1/2 is about 7-15 hours, depending on many factors, including the pH of urine (with an acceleration of the half-life at lower values). Imax - 15-70 minutes. Lethal dosage of d-isomer of amphetamine has not been studied; however, there are theoretical conclusions about large values of LD 50 of 5-10% from the available extrapolated data of the standard racemate of amphetamine sulfate (98-130 mg/kg). Amphetamine is eliminated in almost the same qualitative indicators of known metabolites, however, volume of hippuric acid, benzoyl glycuronide and norephedrine is significantly less (60% of the total data), which is due to the specificity of this racemate in the context of metabolism at the level of deamination. In studies on rodents, a pronounced decrease in genotoxicity was proved in contrast to the standard amphetamine racemate in dose of 4600 mg/ml.
Mechanism of action.
The pharmacodynamic mechanism of action almost does not differ from the fundamental form of amphetamine. Through inhibition and reversion of serotonin, norepinephrine and dopamine transporters, interaction with TAAR1, which ultimately leads to an increase in the concentration of monoamines and catecholamines simultaneously in the synaptic cleft (the gap is 10-50 nm wide between the membranes, the edges are strengthened by intercellular contacts). An important feature of dextroamphetamine specifically, is the fact that the affinity of the right-sided enantiomer with TAAR1 is much greater (by 15-30% according to various sources), and VMAT2 captures a smaller amount of this enantiomer, and the minimum affinity for the serotonin transporter, in contrast to the left-sided one, and the load of such systems as CDK5R1, DLG1 and CAMKIIAA is significantly less with equivalent clinical effects.
No less important is the scientific fact identified by Novascreen BioSciences Corporation that due to lower affinity for a number of receptors (a1, a2, b-adrenoreceptors, mu-opioid, histamine, CCK, NK, GABA, ETeA, and others), the severity of both undesirable side clinical effects and other negative consequences on the molecular level are significantly lower, which determines the priority of this particular form of the substance with the preservation of the corresponding desired clinical effects. The net effect of these multiple action properties is that d-amphetamine increases the "availability" or concentration ability of catecholamines in the extracellular space by reversing the transport of catecholamines from the terminal end of the neuron, which is also one of the features of the right-rotating enantiomer. The indicators of inhibition of the uptake of [3 H] by rat brain synaptosomes by the d-enantiomer vary and differ significantly from amphetamine racemate. For norepinephrine, Ki = nM at a value of 45-50, for dopamine 82-200, for 5-HT, the indicators vary from 1840 to 3830 in rat brain models. A more "soft" effect of the cycle of addiction from this form of amphetamine is a consequence of a relatively lower overexpression of FosB in the nucleus accumbens, the presence of positive effects and the absence of inhibition of CREB by protein and serine phosphotases precisely due to lower affinity and influence on the glutamateergic and serotonin-ergic systems. Neurochemical effects in comparison also have a distinctive ability; in the Heal and Wickens studies, changes in motor activity were detected, where its maximum value was 3-4 times less than with amphetamine hydrochloride or sulfate intake, which is directly related to the rate and molecular correction of dopamine outflow in the striatum of the brain.

Although in vitro experiments provide a good insight into individual mechanisms, the efficacy of amphetamine relative to other indirect monoamine agonists, for example classical reuptake inhibitors, can only be estimated from in vivo experiments. We have used dual-probe intracerebral microdialysis to explore the in vivo effects of d- and l-amphetamine in the spontaneously hypertensive rat (SHR), which has been proposed as a rodent model of ADHD. Both isomers of amphetamine dose-dependently increased the extracellular concentrations of noradrenaline in the prefrontal cortex (PFC) and dopamine in the striatum. The pharmacodynamics of their effects are typical of those reported for monoamine releasing agents, i.e. a fast onset of action with peak increases of noradrenaline and dopamine efflux occurring at 30–45 min, large effects (400–450% of baseline for noradrenaline and 700–1500% of baseline for dopamine), with a relatively rapid decline after the maximum. Although no comparative results have been included in this review, the magnitude of the increases produced by amphetamine’s isomers are greater than those reported for classical reuptake inhibitors such as atomoxetine or bupropion, and there is no dose-effect ceiling to amphetamine’s actions.
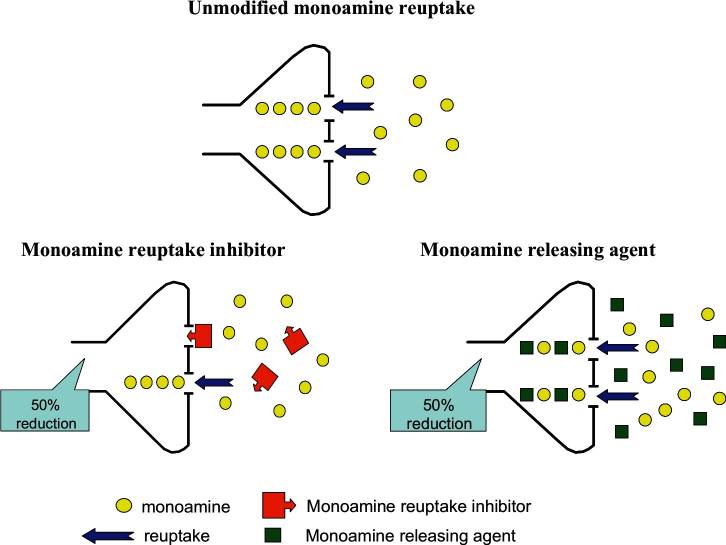
When comparing the effects of drugs on the efflux of catecholamines in the PFC, it is important to consider the highly unusual neuroanatomy of this brain region. The density of DAT sites on PFC dopaminergic neurones is very low, and as a consequence, most dopamine that is released is sequestered via NET into noradrenergic neurones. Even though that there are few DAT sites on PFC dopaminergic neurones, their reuptake capacity is sufficient for amphetamine to evoke substantial dopamine release from them, though it has been suggested that much of the release of dopamine in the PFC comes from noradrenergic neurones. In the Shire Development studies on in vivo models, a statistically significant difference was proved. D-AMP influenced the cognitive components of impulsive control differently: the proportion of impulsive choice decreased with the same level of reward, which also confirms the effectiveness of this enantiomer.
Methods of use and doses.
Recreational dosage of amphetamine d-enantiomer starts from 40-60 mg with oral use, in cases of present tolerance the starting dosage can be increased up to 120-160 mg with a single oral use. When administered intranasally, it is recommended to use a starting dose of 30-50 mg. Parenteral administration of this form is allowed only in case of obtaining a pure solution. Any tablets or powders are recommended for use only by oral and intranasal administration. With parenteral administration, the starting dose is 0,15-0,24 mg/kg.
Clinical presentation of amphetamine intoxication.
The symptoms above are "mild", their severity is up to 40% less than that of the combined form. Therefore, such negative undesirable effects as urinating difficulty, heart rhythm disturbances, increased heart rate, increased blood pressure, increased sweating, high body temperature, nausea, pupil dilation occur exclusively at high doses, and are proportionally different in comparison with racemate.
1. Euphoria and a feeling of increased energy;
2. Prolonged wakefulness;
3. Grandiose ideas or actions;
4. Violent or aggressive behavior;
5. Confirmation bias;
6. Mood lability;
7. Behavior characterized by repeated stereotypes;
8. Auditory, visual or tactile illusions;
9. Personality distortion;
10. Appetite suppression.
Also, people who use D-AMP must have at least two of the following signs to be diagnosed:
1. Tachycardia;
2. Arterial hypertension;
3. Sweating and feverish chills;
4. Nausea or vomiting;
5. Muscle weakness;
6. Pain or discomfort behind the sternum;
7. Psychomotor agitation;
8. Pupil dilation.
Symptoms of D-AMP intoxication mainly include signs of cardiovascular and nervous system complications. Respiratory disorders develop less often.
Cardiac disorders: hypertension and tachycardia are the most common ones, when taking low doses. Initial bradycardia is also possible due to an increase in parasympathetic tone. Arrhythmias (supraventricular are frequent) can be caused by a sympathetic crisis or a blockade of fast sodium channels. Ischemia and myocardial infarction are caused by coronary spasm, which reaches a maximum 30 minutes after intranasal D-AMP use and coincides with the peak concentration of the drug in blood. Further coronary spasm can develop after 90 minutes, which is associated with the accumulation of D-AMP metabolites. An increase in platelet aggregation leads to formation of blood clots in coronary vessels. Moreover, the myocardial oxygen demand increases. A rare complication is aortic rupture.
Special instructions, interactions with other substances.
The absolute contraindications of the use of any form of amphetamine are: severe vascular atherosclerosis, any symptomatic cardiovascular diseases, MAO intake (and 14 days after the last act of using the drug of this group), glaucoma, hyperthyroidism.
The “non-ADIOS" rule:
Non-Alcohol - it is not recommended using with alcohol.
Non-Dissociatives - it is not recommended using with dissociative drugs.
Non-iMAO - it is not recommended using with monoamine oxidase inhibitors.
Non-Opiates - it is not recommended using with opioid receptor agonists.
Non-Stimulators - it is not recommended using with stimulants.
1. When using D-AMP together with drugs of the PDE-5 inhibitor group, the risk of developing side effects of both substances increases. Most common side effects: severe headache of a spastic nature, nausea. Incidence of developing severe critical conditions is low.
2. When using D-AMP together with marijuana, there is a risk of occurrence of increased blood pressure, headache, fine tremor, dryness of the mouth. The probability of developing severe critical conditions is low.
3. When using D-AMP with neuroleptics/tranquilizers, there is a high risk of leveling the main effects of D-AMP. The probability of developing severe critical conditions is low (in cases where there are no contraindications to taking neuroleptics).
4. It is not recommended to use D-AMP during treatment with antibacterial, antiviral and antifungal drugs, hormones, anticoagulants.
5. It is not recommended to use D-AMP during any surgical manipulation and in time before and after that for a certain period (the period is identified individually).
6. It is not recommended to use D-AMP if diagnosed with any disorder of cardiovascular system, kidneys, liver, lungs, central nervous system.
7. It is not recommended to use D-AMP during pregnancy and lactation.
In two earlier published studies, Jasinski and Krishnan compared the subjective effects of IR d-amphetamine in drug-experienced human volunteers when these compounds were administered intravenously and orally. In the trial where they compared these compounds after oral administration, IR d-amphetamine (40 mg (29.6 mg d-amphetamine base)) evoked a statistically significant increase relative to placebo in ‘Drug liking’ on the Drug Rating Questionnaire – Subject (DQRS) scale, whereas the equivalent dose of d-amph (100 mg, oral) did not. Furthermore, the time of d-amph peak pharmacological effect was substantially delayed compared with IR d-amphetamine, at 3.0 h versus 1.5–2.0 h. When d-amfetamine was given at an increased dose of 150 mg, it significantly increased the DQRS ‘Drug liking’ score to an equivalent extent to IR d-amphetamine (40 mg oral). However, the peak effect of the higher dose of d-amfetamine was even more delayed, at 4.0 h. When the intravenous route was explored, IR d-amphetamine (20 mg intravenous) produced a peak ‘Drug liking’ score 20 min after dosing, which coincided with plasma Cmax. In contrast, the equivalent dose of d-amph (50 mg intravenous) did not significantly increase ‘Dug liking’ relative to placebo, and the Cmax of plasma d-amphetamine occurred considerably later at 2.0 h. Both compounds yielded equivalent AUC0-24h values, but compared with the equivalent dose of IR d-amphetamine, the Cmax for plasma d-amphetamine was threefold smaller for lisdexamfetamine and the tmax was threefold greater.
First aid in case of overdose.
The mechanism of toxicity is primarily related to excessive extracellular dopamine, norepinephrine, and serotonin. The primary clinical syndrome involves prominent neurological and cardiovascular effects, but secondary complications can involve renal, muscle, pulmonary, and GI effects. Hyperactivity, hyperthermia, tachycardia, tachypnea, mydriasis, tremors, seizures, and altered mental status are some of the most common signs and symptoms of amphetamine intoxication. Diagnosis can be confirmed by detecting amphetamine in stomach contents or vomitus or by positive urine toxicology for illicit drugs. False-positive amphetamine screen can present following trazodone overdose or bupropion overdose. There is no antidote for amphetamine toxicity; however, activated charcoal is an emergency treatment. In patients who can drink safely, the recommendation to administer activated charcoal, 1 to 2 g/kg up to 100 g by mouth if the ingestion occurred within the previous hour. Amphetamine-related toxicity requires management by controlling life-threatening central nervous system and cardiovascular signs in a quiet environment.
Indications for going to the hospital or calling an ambulance: impaired or absent consciousness, impaired speech, motor activity, lack of orientation in space and time, severe pain behind the sternum lasting more than half an hour, an increase in body temperature of more than 38.0 C or hyperthermia lasting more than half an hour, an increase in blood pressure of more than 180/110 mmHg with no effect of hypotensive therapy.
1. The treatment of patients with blood pressure of more than 140/95 mm Hg includes one tablet of a beta-blocker without intrinsic sympathomimetic activity, one tablet of an ACE inhibitor, after 30 minutes - one tablet of a tranquilizer (0.25 mg of alprazolam).
2. In patients with intense anxiety, panic attack, psychosis: one tablet of a tranquilizer and one tablet of a neuroleptic with a sedative effect, psychological help, emergency psychotherapy.
3. In patients with sternum pain, discomfort in the chest: one tablet of slow calcium channel blockers of the third generation, reflexively reducing the heart rate, one tablet of an ACE inhibitor of the 3rd generation OR one tablet of an agonist of imidazoline effects OR one tablet ; if the pain syndrome does not become less intense within 20 minutes, then it is recommended to go to the hospital.
4. When there is an increase in body temperature of no more than 37.5 C, dynamic observation for half an hour is ordered. Pharmacological treatment is not required. If hyperthermia persists for more than half of an hour (in the absence of external causes), then it is recommended to go to the hospital.
5. In patients with intense headaches, it is recommended to use antispasmodics in combination with sedative herbal remedies or tranquilizers in low doses. If the headache is associated with vomiting, an intramuscular injection of metoclopramide 2.0 ml is recommended. Isolated nausea and functional dyspepsia do not require pharmacological treatment.
6. For tremors, moderate convulsions or mild psychomotor agitation, tranquilizers are recommended. It is strongly advised not to use neuroleptics in these cases.
A beta-blocker such as propranolol can help to manage cardiac tachyarrhythmias. Consider intravenous nitroprusside (start at 0.5 to 1 mcg/kg per minute and titrate as needed) for severe hypertension. Intravenous fluid should be given as it counters hyperthermia, assists in maintaining renal function, and helps promote the elimination of amphetamine and its analogs. In cases of severe agitation, clinicians should consider aggressive treatment to avoid malignant hypertension, rhabdomyolysis, hyperthermia, and seizures. Evidence supports using large doses of benzodiazepines to treat amphetamine-overdose-related psychosis and agitation. In cases where agitation, delirium, and movement disorders are unresponsive to benzodiazepines, second-line therapies include antipsychotics such as ziprasidone or haloperidol, central alpha-adrenoreceptor agonists such as dexmedetomidine, or propofol can be administered. Neuromuscular paralysis, intubation, and active cooling measures may be necessary in severe cases. In patients with tachycardia, obtain ECG, and consider telemetry. Use intravenous fluid and sedation to control cardiac symptoms. In cases of severe hypertension, consider intravenous nitroprusside. Starting 0.9% normal saline and monitoring creatine kinase (CK), electrolytes, and creatinine is the best way to manage rhabdomyolysis.
There are case reports regarding the Takotsubo cardiomyopathy (TTC), also known as stress-induced cardiomyopathy, which is triggered by amphetamine overdose. In one case, a patient presented to the emergency department after ingesting 30 amphetamine salt tablets with symptoms of chest pain and shortness of breath. At the time of presentation, cardiac enzymes were elevated, the electrocardiogram was unremarkable, and ejection fraction (EF) was 25% to 30% with severe hypokinesis. However, 24 hours later, symptoms were resolved, and a repeated echocardiogram performed three days later showed an EF of 60% with no regional wall motion abnormalities.
Terms of detection and storage rules.
As a rule, storage of amphetamine racemate as well as d-enantiomer should be carried out in a place protected from light and moisture, preferably at room temperature or at a temperature of at least 5 C and not more than 40 C. It can be stored in food paper or in polyethylene terephthalate packages without metal contents.
After single intranasal use of dextroamphetamine, the detection period in the urine when using express methods of research is approximately 46-72 hours, with oral use up to 120-220 hours. Prolonged systematic use of dextroamphetamine has not been studied in the context of the identification of metabolites, although, theoretically, it is known that there is a high chance of detecting the main metabolites by PFIA or chromatography-mass spectrometry during 1.5-2 months after the last use of amphetamine (which is less than that of amphetamine racemate). The detection period of dextroamphetamine metabolites in hair is up to 2,5-3 months.
Synthesis of Dextroamphetamine from P2P
Physico-chemical properties and forms of the substance.
Fundamentally, dextroamphetamine, as well as amphetamine racemate, has the appearance of a colorless volatile oily liquid with a characteristic "fishy" smell and is well-soluble in organic solvents. Its boiling point is 200-203 °C. The enantiomer can have a powdery appearance with the formation of small "stones" of white or light yellow/cream color, with a weak smell, weakly hygroscopic, partially soluble in water when it is in the form of a tablet. It is most often found in the form of sulfate, hydrochloride, sucrate and very rarely in the form of phosphate. As a rule, dextroamphetamine is part of various medications such as «Aderral» and «Dexedrine», where it is found in the form of sucrate and sulfate in concentrations of 25% of the total volume. It also is part of «Vyvanse», «Elvanse», «Tyvense», where it is in the proactive state of dimesylate lysdexamphetamine; fermentation with l-lysine, metabolizes it into the active form of d-amphetamine; «Dexedrine». Other trade names of drugs include: Dexedrine sulphate, Afatin, d-Amfetasul, Domafate Obesedrin, Dexten, Maxiton, Sympamin, Simpamina-D, Albemap, Dadex, Ardex, Dexalone, Amsustain, Betafedrina, d-Betaphedrine, Diocurb, Dextrostat.
Pharmacokinetics.
When administered, amphetamine is metabolized by hydroxylation, where the main cytochrome is CYP2D6, beta-hydroxylation (DBH) and oxidative deamination (FMO3); there are other enzymes that are less involved in metabolism of amphetamine, such as butyrate-CoA ligase and glycine-N-acyltransferase. Bioavailability (with unchanged gastrointestinal pH) reaches 70-75% for racemate and up to 80-83% for dextroamphetamine. The binding with plasma proteins reaches 15-30% according to various sources in healthy people, the distribution volume is moderate, the substance actively passes through the blood-brain barrier with a variable coefficient of 17p. T1/2 is about 7-15 hours, depending on many factors, including the pH of urine (with an acceleration of the half-life at lower values). Imax - 15-70 minutes. Lethal dosage of d-isomer of amphetamine has not been studied; however, there are theoretical conclusions about large values of LD 50 of 5-10% from the available extrapolated data of the standard racemate of amphetamine sulfate (98-130 mg/kg). Amphetamine is eliminated in almost the same qualitative indicators of known metabolites, however, volume of hippuric acid, benzoyl glycuronide and norephedrine is significantly less (60% of the total data), which is due to the specificity of this racemate in the context of metabolism at the level of deamination. In studies on rodents, a pronounced decrease in genotoxicity was proved in contrast to the standard amphetamine racemate in dose of 4600 mg/ml.
Mechanism of action.
The pharmacodynamic mechanism of action almost does not differ from the fundamental form of amphetamine. Through inhibition and reversion of serotonin, norepinephrine and dopamine transporters, interaction with TAAR1, which ultimately leads to an increase in the concentration of monoamines and catecholamines simultaneously in the synaptic cleft (the gap is 10-50 nm wide between the membranes, the edges are strengthened by intercellular contacts). An important feature of dextroamphetamine specifically, is the fact that the affinity of the right-sided enantiomer with TAAR1 is much greater (by 15-30% according to various sources), and VMAT2 captures a smaller amount of this enantiomer, and the minimum affinity for the serotonin transporter, in contrast to the left-sided one, and the load of such systems as CDK5R1, DLG1 and CAMKIIAA is significantly less with equivalent clinical effects.
No less important is the scientific fact identified by Novascreen BioSciences Corporation that due to lower affinity for a number of receptors (a1, a2, b-adrenoreceptors, mu-opioid, histamine, CCK, NK, GABA, ETeA, and others), the severity of both undesirable side clinical effects and other negative consequences on the molecular level are significantly lower, which determines the priority of this particular form of the substance with the preservation of the corresponding desired clinical effects. The net effect of these multiple action properties is that d-amphetamine increases the "availability" or concentration ability of catecholamines in the extracellular space by reversing the transport of catecholamines from the terminal end of the neuron, which is also one of the features of the right-rotating enantiomer. The indicators of inhibition of the uptake of [3 H] by rat brain synaptosomes by the d-enantiomer vary and differ significantly from amphetamine racemate. For norepinephrine, Ki = nM at a value of 45-50, for dopamine 82-200, for 5-HT, the indicators vary from 1840 to 3830 in rat brain models. A more "soft" effect of the cycle of addiction from this form of amphetamine is a consequence of a relatively lower overexpression of FosB in the nucleus accumbens, the presence of positive effects and the absence of inhibition of CREB by protein and serine phosphotases precisely due to lower affinity and influence on the glutamateergic and serotonin-ergic systems. Neurochemical effects in comparison also have a distinctive ability; in the Heal and Wickens studies, changes in motor activity were detected, where its maximum value was 3-4 times less than with amphetamine hydrochloride or sulfate intake, which is directly related to the rate and molecular correction of dopamine outflow in the striatum of the brain.
Although in vitro experiments provide a good insight into individual mechanisms, the efficacy of amphetamine relative to other indirect monoamine agonists, for example classical reuptake inhibitors, can only be estimated from in vivo experiments. We have used dual-probe intracerebral microdialysis to explore the in vivo effects of d- and l-amphetamine in the spontaneously hypertensive rat (SHR), which has been proposed as a rodent model of ADHD. Both isomers of amphetamine dose-dependently increased the extracellular concentrations of noradrenaline in the prefrontal cortex (PFC) and dopamine in the striatum. The pharmacodynamics of their effects are typical of those reported for monoamine releasing agents, i.e. a fast onset of action with peak increases of noradrenaline and dopamine efflux occurring at 30–45 min, large effects (400–450% of baseline for noradrenaline and 700–1500% of baseline for dopamine), with a relatively rapid decline after the maximum. Although no comparative results have been included in this review, the magnitude of the increases produced by amphetamine’s isomers are greater than those reported for classical reuptake inhibitors such as atomoxetine or bupropion, and there is no dose-effect ceiling to amphetamine’s actions.
When comparing the effects of drugs on the efflux of catecholamines in the PFC, it is important to consider the highly unusual neuroanatomy of this brain region. The density of DAT sites on PFC dopaminergic neurones is very low, and as a consequence, most dopamine that is released is sequestered via NET into noradrenergic neurones. Even though that there are few DAT sites on PFC dopaminergic neurones, their reuptake capacity is sufficient for amphetamine to evoke substantial dopamine release from them, though it has been suggested that much of the release of dopamine in the PFC comes from noradrenergic neurones. In the Shire Development studies on in vivo models, a statistically significant difference was proved. D-AMP influenced the cognitive components of impulsive control differently: the proportion of impulsive choice decreased with the same level of reward, which also confirms the effectiveness of this enantiomer.
Methods of use and doses.
Recreational dosage of amphetamine d-enantiomer starts from 40-60 mg with oral use, in cases of present tolerance the starting dosage can be increased up to 120-160 mg with a single oral use. When administered intranasally, it is recommended to use a starting dose of 30-50 mg. Parenteral administration of this form is allowed only in case of obtaining a pure solution. Any tablets or powders are recommended for use only by oral and intranasal administration. With parenteral administration, the starting dose is 0,15-0,24 mg/kg.
Clinical presentation of amphetamine intoxication.
The symptoms above are "mild", their severity is up to 40% less than that of the combined form. Therefore, such negative undesirable effects as urinating difficulty, heart rhythm disturbances, increased heart rate, increased blood pressure, increased sweating, high body temperature, nausea, pupil dilation occur exclusively at high doses, and are proportionally different in comparison with racemate.
1. Euphoria and a feeling of increased energy;
2. Prolonged wakefulness;
3. Grandiose ideas or actions;
4. Violent or aggressive behavior;
5. Confirmation bias;
6. Mood lability;
7. Behavior characterized by repeated stereotypes;
8. Auditory, visual or tactile illusions;
9. Personality distortion;
10. Appetite suppression.
Also, people who use D-AMP must have at least two of the following signs to be diagnosed:
1. Tachycardia;
2. Arterial hypertension;
3. Sweating and feverish chills;
4. Nausea or vomiting;
5. Muscle weakness;
6. Pain or discomfort behind the sternum;
7. Psychomotor agitation;
8. Pupil dilation.
Symptoms of D-AMP intoxication mainly include signs of cardiovascular and nervous system complications. Respiratory disorders develop less often.
Cardiac disorders: hypertension and tachycardia are the most common ones, when taking low doses. Initial bradycardia is also possible due to an increase in parasympathetic tone. Arrhythmias (supraventricular are frequent) can be caused by a sympathetic crisis or a blockade of fast sodium channels. Ischemia and myocardial infarction are caused by coronary spasm, which reaches a maximum 30 minutes after intranasal D-AMP use and coincides with the peak concentration of the drug in blood. Further coronary spasm can develop after 90 minutes, which is associated with the accumulation of D-AMP metabolites. An increase in platelet aggregation leads to formation of blood clots in coronary vessels. Moreover, the myocardial oxygen demand increases. A rare complication is aortic rupture.
Special instructions, interactions with other substances.
The absolute contraindications of the use of any form of amphetamine are: severe vascular atherosclerosis, any symptomatic cardiovascular diseases, MAO intake (and 14 days after the last act of using the drug of this group), glaucoma, hyperthyroidism.
The “non-ADIOS" rule:
Non-Alcohol - it is not recommended using with alcohol.
Non-Dissociatives - it is not recommended using with dissociative drugs.
Non-iMAO - it is not recommended using with monoamine oxidase inhibitors.
Non-Opiates - it is not recommended using with opioid receptor agonists.
Non-Stimulators - it is not recommended using with stimulants.
1. When using D-AMP together with drugs of the PDE-5 inhibitor group, the risk of developing side effects of both substances increases. Most common side effects: severe headache of a spastic nature, nausea. Incidence of developing severe critical conditions is low.
2. When using D-AMP together with marijuana, there is a risk of occurrence of increased blood pressure, headache, fine tremor, dryness of the mouth. The probability of developing severe critical conditions is low.
3. When using D-AMP with neuroleptics/tranquilizers, there is a high risk of leveling the main effects of D-AMP. The probability of developing severe critical conditions is low (in cases where there are no contraindications to taking neuroleptics).
4. It is not recommended to use D-AMP during treatment with antibacterial, antiviral and antifungal drugs, hormones, anticoagulants.
5. It is not recommended to use D-AMP during any surgical manipulation and in time before and after that for a certain period (the period is identified individually).
6. It is not recommended to use D-AMP if diagnosed with any disorder of cardiovascular system, kidneys, liver, lungs, central nervous system.
7. It is not recommended to use D-AMP during pregnancy and lactation.
In two earlier published studies, Jasinski and Krishnan compared the subjective effects of IR d-amphetamine in drug-experienced human volunteers when these compounds were administered intravenously and orally. In the trial where they compared these compounds after oral administration, IR d-amphetamine (40 mg (29.6 mg d-amphetamine base)) evoked a statistically significant increase relative to placebo in ‘Drug liking’ on the Drug Rating Questionnaire – Subject (DQRS) scale, whereas the equivalent dose of d-amph (100 mg, oral) did not. Furthermore, the time of d-amph peak pharmacological effect was substantially delayed compared with IR d-amphetamine, at 3.0 h versus 1.5–2.0 h. When d-amfetamine was given at an increased dose of 150 mg, it significantly increased the DQRS ‘Drug liking’ score to an equivalent extent to IR d-amphetamine (40 mg oral). However, the peak effect of the higher dose of d-amfetamine was even more delayed, at 4.0 h. When the intravenous route was explored, IR d-amphetamine (20 mg intravenous) produced a peak ‘Drug liking’ score 20 min after dosing, which coincided with plasma Cmax. In contrast, the equivalent dose of d-amph (50 mg intravenous) did not significantly increase ‘Dug liking’ relative to placebo, and the Cmax of plasma d-amphetamine occurred considerably later at 2.0 h. Both compounds yielded equivalent AUC0-24h values, but compared with the equivalent dose of IR d-amphetamine, the Cmax for plasma d-amphetamine was threefold smaller for lisdexamfetamine and the tmax was threefold greater.
First aid in case of overdose.
The mechanism of toxicity is primarily related to excessive extracellular dopamine, norepinephrine, and serotonin. The primary clinical syndrome involves prominent neurological and cardiovascular effects, but secondary complications can involve renal, muscle, pulmonary, and GI effects. Hyperactivity, hyperthermia, tachycardia, tachypnea, mydriasis, tremors, seizures, and altered mental status are some of the most common signs and symptoms of amphetamine intoxication. Diagnosis can be confirmed by detecting amphetamine in stomach contents or vomitus or by positive urine toxicology for illicit drugs. False-positive amphetamine screen can present following trazodone overdose or bupropion overdose. There is no antidote for amphetamine toxicity; however, activated charcoal is an emergency treatment. In patients who can drink safely, the recommendation to administer activated charcoal, 1 to 2 g/kg up to 100 g by mouth if the ingestion occurred within the previous hour. Amphetamine-related toxicity requires management by controlling life-threatening central nervous system and cardiovascular signs in a quiet environment.
Indications for going to the hospital or calling an ambulance: impaired or absent consciousness, impaired speech, motor activity, lack of orientation in space and time, severe pain behind the sternum lasting more than half an hour, an increase in body temperature of more than 38.0 C or hyperthermia lasting more than half an hour, an increase in blood pressure of more than 180/110 mmHg with no effect of hypotensive therapy.
1. The treatment of patients with blood pressure of more than 140/95 mm Hg includes one tablet of a beta-blocker without intrinsic sympathomimetic activity, one tablet of an ACE inhibitor, after 30 minutes - one tablet of a tranquilizer (0.25 mg of alprazolam).
2. In patients with intense anxiety, panic attack, psychosis: one tablet of a tranquilizer and one tablet of a neuroleptic with a sedative effect, psychological help, emergency psychotherapy.
3. In patients with sternum pain, discomfort in the chest: one tablet of slow calcium channel blockers of the third generation, reflexively reducing the heart rate, one tablet of an ACE inhibitor of the 3rd generation OR one tablet of an agonist of imidazoline effects OR one tablet ; if the pain syndrome does not become less intense within 20 minutes, then it is recommended to go to the hospital.
4. When there is an increase in body temperature of no more than 37.5 C, dynamic observation for half an hour is ordered. Pharmacological treatment is not required. If hyperthermia persists for more than half of an hour (in the absence of external causes), then it is recommended to go to the hospital.
5. In patients with intense headaches, it is recommended to use antispasmodics in combination with sedative herbal remedies or tranquilizers in low doses. If the headache is associated with vomiting, an intramuscular injection of metoclopramide 2.0 ml is recommended. Isolated nausea and functional dyspepsia do not require pharmacological treatment.
6. For tremors, moderate convulsions or mild psychomotor agitation, tranquilizers are recommended. It is strongly advised not to use neuroleptics in these cases.
A beta-blocker such as propranolol can help to manage cardiac tachyarrhythmias. Consider intravenous nitroprusside (start at 0.5 to 1 mcg/kg per minute and titrate as needed) for severe hypertension. Intravenous fluid should be given as it counters hyperthermia, assists in maintaining renal function, and helps promote the elimination of amphetamine and its analogs. In cases of severe agitation, clinicians should consider aggressive treatment to avoid malignant hypertension, rhabdomyolysis, hyperthermia, and seizures. Evidence supports using large doses of benzodiazepines to treat amphetamine-overdose-related psychosis and agitation. In cases where agitation, delirium, and movement disorders are unresponsive to benzodiazepines, second-line therapies include antipsychotics such as ziprasidone or haloperidol, central alpha-adrenoreceptor agonists such as dexmedetomidine, or propofol can be administered. Neuromuscular paralysis, intubation, and active cooling measures may be necessary in severe cases. In patients with tachycardia, obtain ECG, and consider telemetry. Use intravenous fluid and sedation to control cardiac symptoms. In cases of severe hypertension, consider intravenous nitroprusside. Starting 0.9% normal saline and monitoring creatine kinase (CK), electrolytes, and creatinine is the best way to manage rhabdomyolysis.
There are case reports regarding the Takotsubo cardiomyopathy (TTC), also known as stress-induced cardiomyopathy, which is triggered by amphetamine overdose. In one case, a patient presented to the emergency department after ingesting 30 amphetamine salt tablets with symptoms of chest pain and shortness of breath. At the time of presentation, cardiac enzymes were elevated, the electrocardiogram was unremarkable, and ejection fraction (EF) was 25% to 30% with severe hypokinesis. However, 24 hours later, symptoms were resolved, and a repeated echocardiogram performed three days later showed an EF of 60% with no regional wall motion abnormalities.
Terms of detection and storage rules.
As a rule, storage of amphetamine racemate as well as d-enantiomer should be carried out in a place protected from light and moisture, preferably at room temperature or at a temperature of at least 5 C and not more than 40 C. It can be stored in food paper or in polyethylene terephthalate packages without metal contents.
After single intranasal use of dextroamphetamine, the detection period in the urine when using express methods of research is approximately 46-72 hours, with oral use up to 120-220 hours. Prolonged systematic use of dextroamphetamine has not been studied in the context of the identification of metabolites, although, theoretically, it is known that there is a high chance of detecting the main metabolites by PFIA or chromatography-mass spectrometry during 1.5-2 months after the last use of amphetamine (which is less than that of amphetamine racemate). The detection period of dextroamphetamine metabolites in hair is up to 2,5-3 months.
Last edited:
