2C-B
4-BROMO-2,5-DIMETHOXYPHENETHYLAMINE
View attachment 700
SYNTHESIS: A solution of 100 g of 2,5-dimethoxybenzaldehyde in 220 g nitromethane was treated with 10 g anhydrous ammonium acetate, and heated on a steam bath for 2.5 h with occasional swirling. The deep-red reaction mixture was stripped of the excess nitromethane under vacuum, and the residue crystallized spontaneously. This crude nitrostyrene was purified by grinding under IPA, filtering, and air-drying, to yield 85 g of 2,5-dimethoxy-beta-nitrostyrene as a yellow-orange product of adequate purity for the next step. Further purification can be achieved by recrystallization from boiling IPA.
In a round-bottomed 2 L flask equipped with a magnetic stirrer and placed under an inert atmosphere, there was added 750 mL anhydrous THF, containing 30 g LAH. There was then added, in THF solution, 60 g 2,5-dimethoxy-beta-nitrostyrene. The final solution was a dirty yellow-brown color, and it was kept at reflux temperature for 24 h. After cooling, the excess hydride was destroyed by the dropwise addition of IPA. Then 30 mL 15% NaOH was added to convert the inorganic solids to a filterable mass. The reaction mixture was filtered, and the filter cake washed first with THF and then with MeOH. The combined mother liquors and washings were freed of solvent under vacuum and the residue suspended in 1.5 L H2O. This was acidified with HCl, washed with 3x100 mL CH2Cl2, made strongly basic with 25% NaOH, and reextracted with 4x100 mL CH2Cl2. The pooled extracts were stripped of solvent under vacuum, yielding 26 g of oily residue, which was distilled at 120-130 °C at 0.5 mm/Hg to give 21 g of a white oil, 2,5-dimethoxy-phenethylamine (
2C-H) which picks up carbon dioxide from the air very quickly.
To a well-stirred solution of 24.8 g 2,5-dimethoxyphenethylamine in 40 mL glacial acetic acid, there was added 22 g elemental bromine dissolved in 40 mL acetic acid. After a couple of mins, there was the formation of solids and the simultaneous evolution of considerable heat. The reaction mixture was allowed to return to room temperature, filtered, and the solids washed sparingly with cold acetic acid. This was the hydrobromide salt. There are many complicated salt forms, both polymorphs and hydrates, that can make the isolation and characterization of 2C-B treacherous. The happiest route is to form the insoluble hydrochloride salt by way of the free base. The entire mass of acetic acid-wet salt was dissolved in warm H2O, made basic to at least pH 11 with 25% NaOH, and extracted with 3x100 mL CH2Cl2. Removal of the solvent gave 33.7 g of residue which was distilled at 115-130 °C at 0.4 mm/Hg. The white oil, 27.6 g, was dissolved in 50 mL H2O containing 7.0 g acetic acid. This clear solution was vigorous stirred, and treated with 20 mL concentrated HCl. There was an immediate formation of the anhydrous salt of 2,5-dimethoxy-4-bromophenethylamine hydrochloride (2C-B). This mass of crystals was removed by filtration (it can be loosened considerably by the addition of another 60 mL H2O), washed with a little H2O, and then with several 50 mL portions of Et2O. When completely air-dry, there was obtained 31.05 g of fine white needles, with a mp of 237-239 °C with decomposition. When there is too much H2O present at the time of adding the final concentrated HCl, a hydrated form of 2C-B is obtained. The hydrobromide salt melts at 214.5-215 °C. The acetate salt was reported to have a mp of 208-209 °C.
DOSAGE: 12 - 24 mg.
DURATION: 4 - 8 h.
DOM
STP; 2,5-DIMETHOXY-4-METHYLAMPHETAMINE
View attachment 701
SYNTHESIS: To a solution of 54.9 g 2,5-dimethoxy-4-methylbenzaldehyde (see the recipe for
2C-D for its preparation) in 215 g glacial acetic acid there was added 19.5 g anhydrous ammonium acetate and 30.6 g nitroethane. This mixture was heated for 3 h on the steam bath, the reaction mixture was cooled in a wet ice bath, allowing the spontaneous formation of yellow crystals. As much H2O as possible was added (just short of a persistent cloudy oily character) and after a few additional h standing, the crystalline 1-(2,5-dimethoxy-4-methylphenyl)-2-nitropropene was removed by filtration and recrystallized from boiling acetic acid. The yield, after drying to constant weight, was 28.3 g and the mp was 87-88 °C. Anal. (C12H15NO4) C, H ,N.
A suspension of 9.5 g LAH in 750 mL well stirred anhydrous Et2O was held at reflux under an inert atmosphere, with the return of the condensed solvent passing through a Soxhlet thimble containing 9.5 g 1-(2,5-dimethoxy-4-methylphenyl)-2-nitropropene. After the addition of the nitrostyrene was complete, the stirred suspension was maintained at reflux for an additional 4 h, then cooled to room temperature and allowed to continue stirring overnight. The excess hydride was destroyed by the addition of 750 mL 8% H2SO4, cautiously, until the hydrogen evolution ceased, then at a speed that allowed the formed solids to disperse. The phases were separated, the aqueous phase washed once with Et2O, treated with 225 g potassium sodium tartrate, and finally made basic (pH >9) with 5% NaOH. This was extracted with 3x150 mL CH2Cl2, the extracts pooled, and the solvent removed under vacuum. The residue was 9.6 g of a clear oil which spontaneously formed crystals with a mp of 60.5-61 °C from hexane. These solids were dissolved in 150 mL anhydrous Et2O, and saturated with anhydrous HCl gas. After standing at room temperature for 2 h, the crystalline 2,5-dimethoxy-4-methylamphetamine hydrochloride (DOM) was removed by filtration, washed with Et2O, and air dried to constant weight. There was obtained 8.25 g of glistening white crystals that had a mp of 190.5-191.5 °C. The sulfate had a mp of 131 °C. Anal. (C12H20ClNO2) C, H ,N.
The above nitrostyrene may also be converted to the final amine product through the intermediary of the corresponding phenylacetone. To a well stirred suspension of 10.4 g powdered iron in 20 mL glacial acetic acid held at reflux temperature, there was added 4.9 g 1-(2,5-dimethoxy-4-methylphenyl)-2-nitropropene as a solid. Refluxing was continued for 2 h and then all was filtered through wet Celite. After washing with 300 mL H2O followed by 300 mL Et2O, the combined filtrate and washes were separated, and the aqueous phase extracted with 2x100 mL Et2O. The organic phase and extracts were combined and washed with 2x100 mL saturated K2CO3 and the solvent was removed under vacuum yielding a reddish oil weighing 3.3 g. This was distilled at 111-115 °C at 0.5 mm/Hg to give a pale green solid. After recrystallization from benzene, there was obtained 2.8 g 1-(2,5-dimethoxy-4-methylphenyl)-2-propanone as white crystals with a mp of 57-59 °C. This ketone has also been described as a pale-yellow oil with a bp of 115-118 °C at 0.4 mm/Hg. A solution of 0.7 g 1-(2,5-dimethoxyphenyl-4-methyl)-2-propanone in 20 mL MeOH was treated with 6.0 g ammonium acetate, 0.3 g sodium cyanoborohydride, and 3 g Linde 3 A molecular sieves. The mixture was stirred overnight, the solids removed by filtration, and the filtrate dissolved in 100 mL H2O. The solution was acidified with dilute H2SO4, and washed with 2x25 mL CH2Cl2. The aqueous phase was made basic with aqueous NaOH, and the product extracted with 2x25 mL CH2Cl2. The solvent was removed under vacuum, and the residue distilled (at 160 °C at 0.2 mm/Hg) to give colorless product which was dissolved in 3 mL IPA, neutralized with concentrated HCl, and diluted with 50 mL anhydrous Et2O. There was obtained 0.18 g of 2,5-dimethoxy-4-methylamphetamine hydrochloride (DOM) as a white solid with a mp of 187-188 °C.
The optical isomers of DOM have been prepared in two ways. The racemic base has been resolved as the ortho-nitrotartranilic acid salt by recrystallization from EtOH. The (+) acid provides the (+) or "S" isomer of DOM preferentially. Also, the above-mentioned 1-(2,5-dimethoxy-4-methylphenyl)-2-propanone can be reductively aminated with optically active alpha-methyl benzylamine with Raney Nickel. This amine is isolated and purified by recrystallization of the hydrochloride salt. When optically pure, the benzyl group was removed by hydrogenolysis with palladium on carbon. The mp of either of the optical isomers, as the hydrochloride salts, was 204-205 °C.
DOSAGE: 3 - 10 mg.
DURATION: 14 - 20 h.
MDA
3,4-METHYLENEDIOXYAMPHETAMINE
View attachment 702
SYNTHESIS: (from piperonal) To a solution of 15.0 g piperonal in 80 mL glacial acetic acid there was added 15 mL nitroethane followed by 10 g cyclohexylamine. The mixture was held at steam-bath temperature for 6 h, diluted with 10 mL H2O, seeded with a crystal of product, and cooled overnight at 10 °C. The bright yellow crystals were removed by filtration, and air dried to yield 10.7 g of 1-(3,4-methylenedioxyphenyl)-2-nitropropene with a mp of 93-94 °C. This was raised to 97-98 °C by recrystallization from acetic acid. The more conventional efforts of nitrostyrene synthesis using an excess of nitroethane as a solvent and anhydrous ammonium acetate as the base, gives impure product in very poor yields. The nitrostyrene has been successfully made from the components in cold MeOH, with aqueous NaOH as the base.
A suspension of 20 g LAH in 250 mL anhydrous THF was placed under an inert atmosphere and stirred magnetically. There was added, dropwise, 18 g of 1-(3,4-methylenedioxyphenyl)-2-nitropropene in solution in THF and the reaction mixture was maintained at reflux for 36 h. After being brought back to room temperature, the excess hydride was destroyed with 15 mL IPA, followed by 15 mL of 15% NaOH. An additional 50 mL H2O was added to complete the conversion of the aluminum salts to a loose, white, easily filtered solid. This was removed by filtration, and the filter cake washed with additional THF. The combined filtrate and washes were stripped of solvent under vacuum, and the residue dissolved in dilute H2SO4. Washing with 3x75 mL CH2Cl2 removed much of the color, and the aqueous phase was made basic and reextracted with 3x100 mL CH2Cl2. Removal of the solvent yielded 13.0 g of a yellow-colored oil that was distilled. The fraction boiling at 80-90 °C at 0.2 mm weighed 10.2 g and was water-white. It was dissolved in 60 mL of IPA, neutralization with concentrated HCl, and diluted with 120 mL of anhydrous Et2O which produced a lasting turbidity. Crystals formed spontaneously which were removed by filtration, washed with Et2O, and air dried to provide 10.4 g of 3,4-methylenedioxyamphetamine hydrochloride (MDA) with a mp of 187-188 °C.
(from 3,4-methylenedioxyphenylacetone) To a solution of 32.5 g anhydrous ammonium acetate in 120 mL MeOH, there was added 7.12 g 3,4-methylenedioxyphenylacetone (see under MDMA for its preparation) followed by 2.0 g sodium cyanoborohydride. The resulting yellow solution was vigorously stirred, and concentrated HCl was added periodically to keep the pH of the reaction mixture between 6 and 7 as determined by external damp universal pH paper. After several days, undissolved solids remained in the reaction mixture and no more acid was required. The reaction mixture was added to 600 mL of dilute HCl, and this was washed with 3x100 mL CH2Cl2. The combined washes were back-extracted with a small amount of dilute HCl, the aqueous phases combined, and made basic with 25% NaOH. This was then extracted with 3x100 mL CH2Cl2, these extracts combined, and the solvent removed under vacuum to provide 3.8 g of a red-colored residue. This was distilled at 80-90 °C at 0.2 mm/Hg to provide 2.2 g of an absolutely water-white oil. There was no obvious formation of a carbonate salt when exposed to air. This was dissolved in 15 mL IPA, neutralized with 25 drops of concentrated HCl, and diluted with 30 mL anhydrous Et2O. Slowly there was the deposition of white crystals of 3,4-methylenedioxyamphetamine hydrochloride (MDA) which weighed 2.2 g and had a mp of 187-188 °C. The preparation of the formamide (a precursor to MDMA) and the acetamide (a precursor to MDE) are described under those entries.
DOSAGE: 80 - 160 mg.
DURATION: 4 - 6 (
revised, Sep 2001).
MESCALINE;
3,4,5-TRIMETHOXYPHENETHYLAMINE
View attachment 976
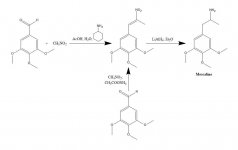
SYNTHESIS: A solution of 20 g 3,4,5-trimethoxybenzaldehyde, 40 mL nitromethane, and 20 mL cyclohexylamine in 200 mL of acetic acid was heated on the steam bath for 1 h. The reaction mixture was then diluted slowly and with good stirring, with 400 mL H2O, which allowed the formation of a heavy yellow crystalline mass. This was removed by filtration, washed with H2O, and sucked as dry as possible. Recrystallization from boiling MeOH (15 mL/g) yielded, after filtration and air drying, beta-nitro-3,4,5-trimethoxystyrene as bright yellow crystals weighing 18.5 g. An alternate synthesis was effective, using an excess of nitromethane as solvent as well as reagent, if the amount of ammonium acetate catalysis was kept small. A solution of 20 g 3,4,5-trimethoxybenzaldehyde in 40 mL nitromethane containing 1 g anhydrous ammonium acetate was heated on the steam bath for 4 h. The solvent was stripped under vacuum and the residual yellow oil was dissolved in two volumes of hot MeOH, decanted from some insolubles, and allowed to cool. The crystals formed are removed by filtration, washed with MeOH and air dried yielding 14.2 g. of bright yellow crystals of beta-nitro-3,4,5-trimethoxystyrene. The use of these proportions but with 3.5 g ammonium acetate gave extensive side-reaction products even when worked up after only 1.5 h heating. The yield of nitrostyrene was, in this latter case, unsatisfactory.
To a gently refluxing suspension of 2 g LAH in 200 mL Et2O, there was added 2.4 g beta-nitro-3,4,5-trimethoxystyrene as a saturated Et2O solution by use of a Soxhlet extraction condenser modified to allow the continuous return of condensed solvent through the thimble. After the addition was complete, the refluxing conditions were maintained for another 48 h. After cooling the reaction mixture, a total of 150 mL of 1.5 N H2SO4 was cautiously added, destroying the excess hydride and untimately providing two clear phases. These were separated, and the aqueous phase was washed once with 50 mL Et2O. There was then added 50 g potassium sodium tartrate, followed by sufficient NaOH to bring the pH >9. This was then extracted with 3x75 mL CH2Cl2, and the solvent from the pooled extracts was removed under vacuum. The residue was distilled at 120-130 °C at 0.3 mm/Hg giving a white oil that was dissolved in 10 mL IPA and neutralized with concentrated HCl. The white crystals that formed were diluted with 25 mL Et2O, removed by filtration, and air dried to provide 2.1 g 3,4,5-trimethoxyphenethylamine hydrochloride (M) as glistening white crystals. The sulfate salt formed spectacular crystals from water, but had a broad and uncharacteristic mp. An alternate synthesis can employ 3,4,5-trimethoxyphenylacetonitrile, as described under beta-D.
DOSAGE: 200-400 mg (as the sulfate salt), 178-356 mg (as the hydrochloride salt)
[Erowid Note: The original text read "178-256" but this was an error. The error was found by Bo and verified with Shulgin. See the Erowid Mescaline Dosage page for a more complete discussion of the forms of mescaline].
DURATION: 10-12 h
TMA
3,4,5-TRIMETHOXYAMPHETAMINE
View attachment 704
SYNTHESIS: To a solution of 39.2 g 3,4,5-trimethoxybenzaldehyde in 30 mL warm EtOH there was added 15.7 g nitroethane followed by 1.5 mL n-butylamine. The reaction mixture was allowed to stand at 40 °C for 7 days. With cooling and scratching, fine yellow needles were obtained which, after removal by filtration and air drying, weighed 48 g. Recrystallization from EtOH gave 2-nitro-1-(3,4,5-trimethoxyphenyl)propene as yellow crystals with a mp of 94-95 °C. Anal. (C12H15NO5) C,H,N. Alternatively, a solution of 20 g of the aldehyde in 75 mL nitroethane was treated with 4 g anhydrous ammonium acetate and heated on the steam bath until a deep red color had been generated. Removal of the excess solvent/reagent under vacuum gave a red oil which was dissolved in an equal volume of boiling MeOH. On cooling, yellow crystals of the nitropropene separated. Recrystallization from MeOH gave, after air drying to constant weight, 13.0 g with the same mp.
Under an inert atmosphere, 38 g LAH was wetted with 100 mL anhydrous Et2O, and then suspended in 1 L dry THF. This was brought up to a gentle reflux, and there was added, slowly, a solution of 43.7 g 2-nitro-1-(3,4,5-trimethoxyphenyl)propene in 160 mL THF. Refluxing was continued for 36 h, and then the reaction mixture was cooled with an external ice bath. The excess hydride was destroyed by the cautious addition of 38 mL H2O, and this was followed by 38 mL 15% NaOH, and finally another 114 mL H2O. The inorganic salts which should have ended up as a loose, granular, easily filterable mass, looked rather like library paste, but they were filtered nonetheless. Washing with THF was attempted, but it was not efficient. The combined filtrate and washes were stripped of solvent under vacuum giving 31.5 g of the crude base as an amber oil. This was dissolved in 140 mL IPA, neutralized with concentrated HCl (15 mL was required), and diluted with 650 mL anhydrous Et2O. There was an initial oily phase which on continued stirring changed to pale pink solids. These were finely ground under CH3CN to give 15.2 g of 3,4,5-trimethoxyamphetamine hydrochloride (TMA) as white crystals that melted at 195-211 °C. All aluminum salts from everywhere were dissolved in dilute HCl, and 1 Kg of potassium sodium tartrate was added. There as added 25% NaOH allowed the pH to bring the pH to >9 without the precipitation of basic alumina. Extraction of this phase with CH2Cl2 was followed by removal of the solvent and salt formation as described above, allowed the isolation of an additional 6.4 g TMA. The product prepared in this manner contains some 10-15% 3,5-dimethoxy-4-hydroxyamphetamine as an impurity. A solution of 20 g of the TMA made in this manner in 200 mL 5% NaOH was extracted with 2x200 mL CH2Cl2. The pooled extracts were washed with 4x100 mL 5% NaOH, and the aqueous washes were pooled with the original base phase. The organic phase was stripped of its CH2Cl2 under vacuum to give an oil that was dissolved in 40 mL IPA, neutralized with concentrated HCl, and diluted with 400 mL anhydrous Et2O. There was the immediate formation of spectacular white crystals of pure 3,4,5-trimethoxyamphetamine hydrochloride, weighing 15.4 g and having a mp of 220-221 °C. The aqueous phase was brought to neutrality, treated with 10 g potassium di-hydrogen phosphate, brought to pH 9.0 with the careful addition of NaOH, and extracted with 5x100 mL CH2Cl2. Evaporation of the solvent under vacuum gave an oil that spontaneously crystallized. This product, 3,5-dimethoxy-4-hydroxyamphetamine could be further purified by
sublimation at 130 °C at 0.2 mm/Hg. It was a white crystalline solid that slowly discolored in the air. The literature describes a picrate salt with a mp of 225 °C from EtOH.
DOSAGE: 100 - 250 mg.
DURATION: 6 - 8 h.