William D.
Expert
- Joined
- Jul 19, 2021
- Messages
- 1,056
- Reaction score
- 1,324
- Points
- 113
Reaction scheme:
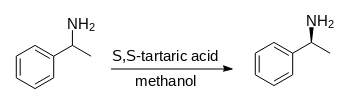
This compound need for dextroamphetamine synthesis.
Synthesis:
1. 124 g (S, S)-tartaric acid in 1800 ml methanol is placed in a flask.
2. Heated to boiling.
3. Slowly dropwise 100 g of racemic α-methylbenzylamine base.
4. Stop warming and left at room temperature for 24 hours.
5. Separated the dropped crystals on filter, and washing with methanol. Crop 75 g.
6. The combined methanol was evaporated to 700 ml amount.
7. The mixture left for 24 hours at room temperature.
8. Separated the dropped crystals on filter, and washing with methanol. Crop 12 g.
9. All separated crystals were combined and dissolved in hot 2000 ml methanol and concentrated to 1400 ml.
10. Repeat steps 4,5,6,7,8 and dry crystals (61 g), concentrated to 300 ml and after 24 hours filtered and dry 13 g crystals.
11. The total yield of the pure (−)-amine salt is 74 g.
This compound need for dextroamphetamine synthesis.
Synthesis:
1. 124 g (S, S)-tartaric acid in 1800 ml methanol is placed in a flask.
2. Heated to boiling.
3. Slowly dropwise 100 g of racemic α-methylbenzylamine base.
4. Stop warming and left at room temperature for 24 hours.
5. Separated the dropped crystals on filter, and washing with methanol. Crop 75 g.
6. The combined methanol was evaporated to 700 ml amount.
7. The mixture left for 24 hours at room temperature.
8. Separated the dropped crystals on filter, and washing with methanol. Crop 12 g.
9. All separated crystals were combined and dissolved in hot 2000 ml methanol and concentrated to 1400 ml.
10. Repeat steps 4,5,6,7,8 and dry crystals (61 g), concentrated to 300 ml and after 24 hours filtered and dry 13 g crystals.
11. The total yield of the pure (−)-amine salt is 74 g.
Last edited by a moderator:
